 
How do your students choose to group the elements? How are they ordering them? What does their table look like? Ask them to use this to arrange and organise the elements into a logical grid structure. Take your students back in time to the 1860s and give them some of the information about the elements that the mid-19th century chemists would have had access to. Scientific ideas can change – models are modified as new evidence is found.There are parallels in the development of the periodic table and the development of our understanding of the atom.The periodic table is a live document, updated and improved as new discoveries are made and evidence found.Patterns in the periodic table help chemists to predict properties of elements.Chemists have been trying to order the elements since they were first discovered.Hearing tales of the early attempts at constructing a periodic table in the 1860s, and about the endeavours of those smashing atoms together today ( rsc.li/35P4bG2) in the quest to extend the periodic table, will also give students an appreciation of how science really works. All the 118 elements were officially proclaimed by the International Union of Pure and Applied Chemistry (IUPAC).Weaving the human stories behind the periodic table into our teaching can help with this. Out of the 118 elements in the periodic table, 94 were discovered naturally and the remaining 24 elements were discovered by laboratory experiments. In 1895, J Thomsen devised a new table interpreted in terms of the electronic structure of atoms by Niels Bohr in 1922. The first and second periods of seven were later distinguished by the use of the letters ‘a’ and ‘b’ attached to the group symbols, which were the Roman numerals. 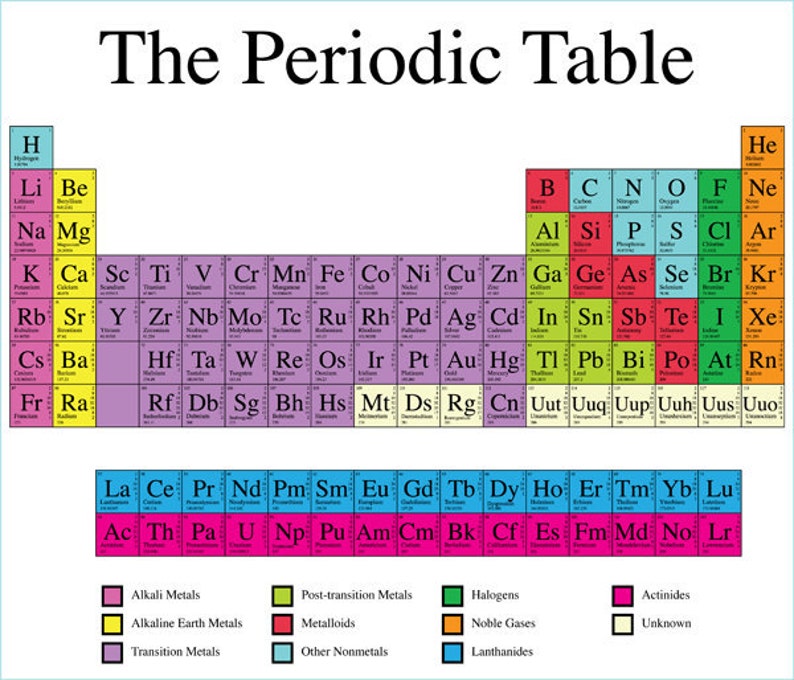
He and Lothar Meyer proposed a table with eight columns obtained by splitting each of the long periods into a period of seven, an eighth group containing the three central elements, and a second period of seven. In 1871, Mendeleyev presented a revision of the 17-group table. It contained 17 columns, with two nearly complete periods of elements, from potassium to bromine and rubidium to iodine, preceded by two partial periods of seven elements each, and followed by three incomplete periods. He presented the table to the Russian Chemical Society on March 6, 1869. Then in 1869, as a result of an extensive correlation of the properties and the atomic weights of the elements, with special attention to valency, Mendeleyev proposed the periodic law. This relationship was termed the law of octaves, by analogy with the seven intervals of the musical scale. According to Britannica, Newlands in 1864 proposed classifying the elements in the order of increasing atomic weights, the elements being assigned ordinal numbers from unity upward and divided into seven groups having properties closely related to the first seven of the elements then known: hydrogen, lithium, beryllium, boron, carbon, nitrogen, and oxygen. He laid the groundwork for the periodic table and later scientists expanded it. In 1817, JW Dobereiner showed that the combining weight, meaning atomic weight, of strontium lies midway between those of calcium and barium. He was inspired by the challenges that scientists had to overcome to develop the modern periodic table and took the initiative to celebrate National Periodic Table Day based upon John Newlands’ first table of the elements published on February 7, 1863. The idea was conceived by David T Steineker, author, inventor, and chemistry teacher at Jefferson County Public Schools in Kentucky. Since 2016, National Periodic Table Day is being celebrated every year on February 7 as a way to pay tribute to the table and the scientists behind its origin. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
